CASE STUDY
A Global Pharmaceutical Company Develops a Data Centric Technology Strategy for R&D
Every year, the pharmaceutical industry develops a variety of new drugs that provide valuable medical benefits, and this has been growing over the years. To further accelerate R&D’s ability to develop new drugs, the right technology, data, and processes need to be in place. As an example, just five years ago, the pharmaceutical technology required to create mRNA vaccines wasn’t available.
The tools needed for breakthrough developments may go beyond existing in-house technology. Therefore, consideration of emerging technologies is important to enabling success. Pharma organizations understand the importance of developing a digital strategy that leverages existing and emerging technologies more efficiently.
A 2020 GlobalData survey found that digital strategies have become a more urgent priority in the healthcare industry, with 30% of companies responding that they’re creating digital strategies, up from 26% in 2019.
To optimize R&D operations both in the short term and into the future, organizations need to ensure they develop an effective strategy and roadmap. This case study discusses how ResultWorks assisted one global pharma’s R&D organization to:
- Define their needed business and technical capabilities
- Design a logical capabilities platform to support the R&D organization
- Develop a reference architecture to support the platforms
- Define a physical platform to support R&D
- Align on a technology strategy and roadmap for execution
Business Challenge
This global pharmaceutical company had grown steadily over many decades through many acquisitions. For much of that period, technology management was decentralized allowing each business unit and site to invest independently. Over time, this led to a significant duplication of systems and processes with inconsistent data.
As executive leadership called for more capabilities to leverage research and development efforts globally, it became clear that the technology strategy needed to change. To fuel the next stage of company growth, they desired to integrate technology capabilities across research and early development functional areas, and across many geographic regions.
How Astrix Enabled Success
Astrix was called upon to develop a comprehensive strategy and roadmap to move to a modern, global, data centric, platform-based architecture with a mix of cloud and on-premises technologies. The strategy also needed to enhance company capabilities for interchange and collaboration with external partners and vendors.
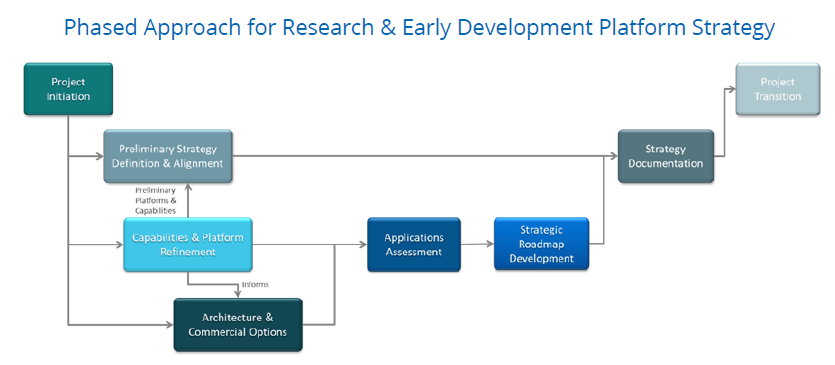
Astrix offered a phased project approach outlined in the graphic below. One of the first hurdles was to transition the team thinking about technology platforms where a platform is defined as a suite of modules/ applications/ components that are used together to deliver multiple complex business processes. Platform definition must be approached logically to model specific domain needs expressed independently of technologies. Only after the logical platform has been defined can the physical platform be addressed considering actual commercial solutions and technologies.
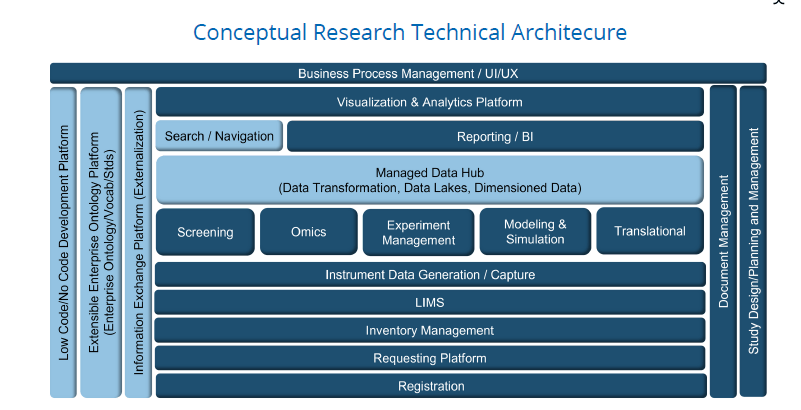
Once business capabilities and logical platforms were defined, the team evolved a technical architecture. A generalized conceptual version of the architecture is shown in the graphic above structured as layers of logical technology platforms. The platforms provided the basis to assess commercial technology options from current vendor offerings to vendor technology plans over the ensuing planning horizon. Over 40 technology vendors were evaluated at a level sufficient to determine the top vendors for each platform. A subsequent applications assessment mapped existing technologies in use by the client to the viable commercial options under consideration. A comprehensive strategy roadmap was then developed for each platform with detailed project descriptions for each identified project.
Fundamental to a program of this size is the management and team commitment to driving through the myriad of organizational, process, data, and technology issues. At the outset, a cohesive team was built, and the team adopted a set of guiding elements, agreed on a vision statement and a purpose, and incorporated key business drivers into the decision making throughout the project. The result was a comprehensive strategy document structured to meet the vision of providing the right data, to the right people, at the right time.
